Welcome to the upcoming double c-myc seminar
- Date: Thursday, August 21
- Time: 13:00 – 14:00
- Location: Birkhaugsalen, D305, 3rd floor, Central Block, Haukeland University Hospital
with the following two presenters:
- 13:00 –Assoc. Prof. Morten Rye, Dept. of Clinical and Molecular Medicine, NTNU
Spatial Transcriptomics Analysis of Prostate Cancer
- 13:20 –Prof. Berit Johansen, Dept. of Biology, NTNU
Treating cancer and leukemia: A new way to trigger cell self-destruction
Presenter: Assoc. Prof. Morten Rye, Department of Clinical and Molecular Medicine, Norwegian University of Science and Technology
Title: Spatial Transcriptomics Analysis of Prostate Cancer
Abstract: The cancerous prostate gland is a heterogenous mix of different cancer and normal cells and tissue types, which is a major source of confounding when large scale molecular – omics analysis is performed on the prostate gland. One way to delineate this heterogeneity is Spatial Transcriptomics, which can generate individual tissue specific gene expression profiles from thousands of spots placed on a grid across a prostate cancer tissue slice (see illustration). A major clinical challenge in prostate cancer is the ability to distinguish aggressive and non-aggressive prostate cancer in the cancer care pathway, and Spatial Transcriptomics could be a technology to do more precise molecular diagnostics of prostate cancer. With this in mind, our Prostomics research group generated spatial transcriptomics datasets (Visium 10x Genomics) on 32 tissue samples from 8 prostate cancer patients, 5 containing aggressive and 3 containing non- aggressive cancers. Samples were taken specifically from cancer, cancer-adjacent and distant normal tissue regions, and spots where classified according to their content of cancer, extracellular matrix/stroma and normal tissue, and further analysed by Gene Set Enrichment Analysis. Our analysis discovered that normal appearing tissue adjacent to aggressive cancer regions lose prostate specific functions and adapt properties resembling cell-types which are not expected to be present in a healthy prostate gland.
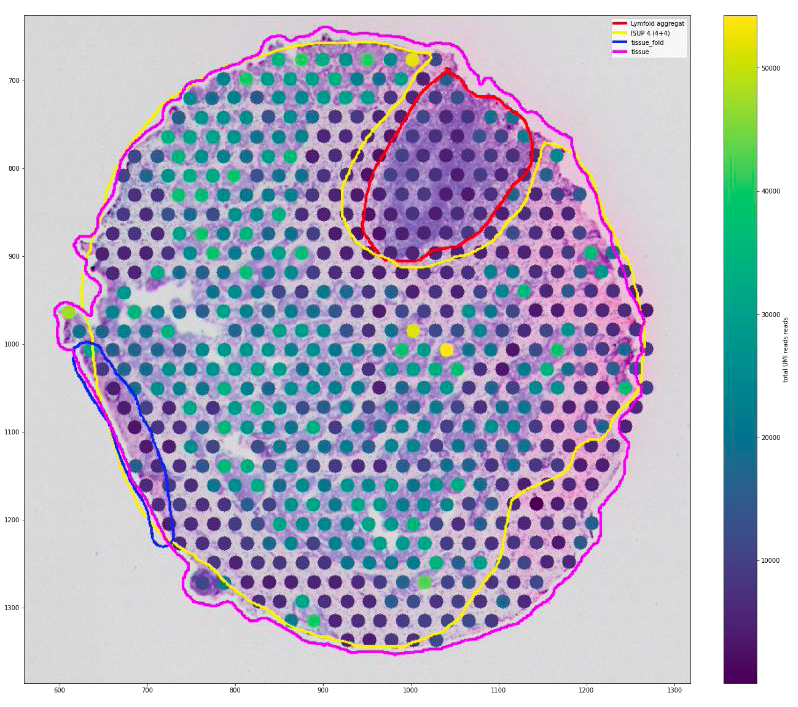
Spatial Transcriptomics on a prostate cancer tissue slice. Each spot generates a transcriptomic profile representative for the cell/tissue content at its location. The marked areas are classification from histopathology.
Presenter: Prof. Berit Johansen, Department of Biology, Norwegian University of Science and Technology
Title: Treating cancer and leukemia: A new way to trigger cell self-destruction
Abstract: The polyunsaturated fatty acid arachidonic acid (AA) and its metabolites, eicosanoids, i.e. prostaglandins, thromboxanes, leukotrienes, etc., have attracted considerable interest in relation to inflammatory processes and diseases, including cancer (leukemia), cardiovascular and autoimmune diseases, and metabolic disorders. AA is generated by the action of phospholipase A2 (PLA2), which catalyzes its hydrolytic cleavage from membrane glycerophospholipids. PLA2 enzymes constitute a superfamily of enzymes, divided into many groups and subgroups. Among them, cytosolic Group IVA PLA2 (cPLA2α) plays a critical role in the release of AA and is the most well-studied PLA2 in inflammation. Targeting eicosanoid production by inhibiting cPLA2α offers a promising approach for inflammation or cancer therapy. Our findings imply cPLA2α may support cancer by mitigating oxidative stress and inhibiting tumor suppressor expression suggesting that the very specific and sensitive cPLA2α -inhibitors we have developed, has potential for treating acute leukemias and other cancers by introducing programmed cell death.
References:
Nur Mahammad, Felicity J. Ashcroft, Astrid J. Feuerherm, Samah Elsaadi, Esten N. Vandsemb, Magne Børset, Berit Johansen. Inhibition of cytosolic phospholipase A2a induces apoptosis in multiple myeloma cells. Molecules, Dec 9;26(24):7447. doi: 10.3390/molecules26247447, 2021
Ashcroft FJ, Bourboula A, Mahammad N, Barbayianni E, Feuerherm AJ, Nguyen TT, Daiki H, Kokotou MG, Kostas A, Dennis EA, Kokotos G, Johansen B. Next generation thiazolyl ketone inhibitors of cytosolic phospholipase A2 α for targeted cancer therapy. Nature Communications. 16(1)124, 2025.
Mahammad N, Feuerherm AJ, Abdollahi P, Børset M, Johansen B, Ashcroft FJ. cPLA2α inhibition leads to RIP1 kinase-dependent cell death in acute leukemia cells. Cell death and disease (in revision for publication) 2025.
